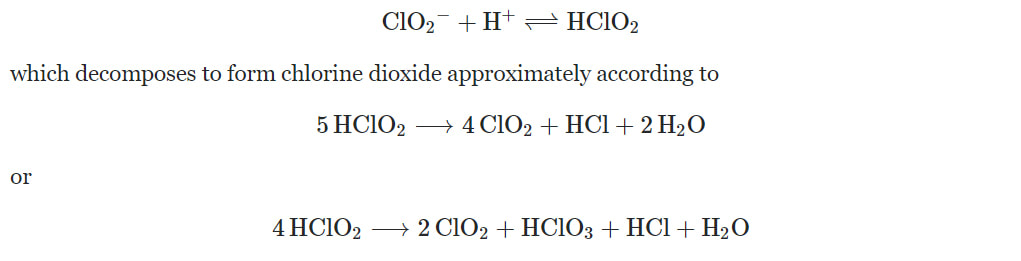True Story of Stabilized Chlorine Dioxide
Most Stabilized Chlorine Dioxide products on the market are purchased in the Sodium Chlorite form and are not actually Chlorine Dioxide. Stabilized Chlorine Dioxide is misleading since the solution does not contain relevant amounts of Chlorine Dioxide. The solution actually contains Sodium Chlorite (NaClO2), which is the salt of the unstable Chlorous acid (HClO2). At low pH, chlorite solutions contain Chlorous Acid which decomposes to form Chlorine Dioxide approximately. At higher pH, decomposition of Sodium Chlorite slows down. Therefore, Sodium Chlorite is mainly stabilized by using a buffered solution. In your product, the buffer consists of trisodium phosphate and citric acid. Thus, a better description would be “stabilized sodium chlorite” instead of “stabilized chlorine dioxide”